Determine sigma and pi bonds1/13/2024  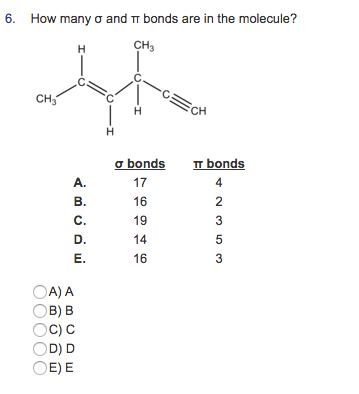
This geometric arrangement makes perfect sense if you consider that it is precisely this angle that allows the four orbitals (and the electrons in them) to be as far apart from each other as possible. ( select ‘load sp 3‘ and ‘load H 1s’ to see orbitals). In the new electron configuration, each of the four valence electrons on the carbon occupies a single sp 3 orbital. In this picture, the four valence orbitals of the carbon (one 2 s and three 2 p orbitals) combine mathematically (remember: orbitals are described by wave equations) to form four equivalent hybrid orbitals, which are called sp 3 orbitals because they are formed from mixing one s and three p orbitals. In order to explain this observation, valence bond theory relies on a concept called orbital hybridization. How does the carbon form four bonds if it has only two half-filled p orbitals available for bonding? A hint comes from the experimental observation that the four C-H bonds in methane are arranged with tetrahedral geometry about the central carbon, and that each bond has the same length and strength. This picture is problematic when it comes to describing the bonding in methane.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
